Details of
UR Pedersen, AK Bacher, TB Schrøder, JC DyreThe EXP pair-potential system. III. Thermodynamic phase diagram,
The Journal of Chemical Physics, 150:174501,2019.
The dataset contains the following parts/files:
Readme file
Pyton script for generating figures
Lattice energy as function of density at T=0
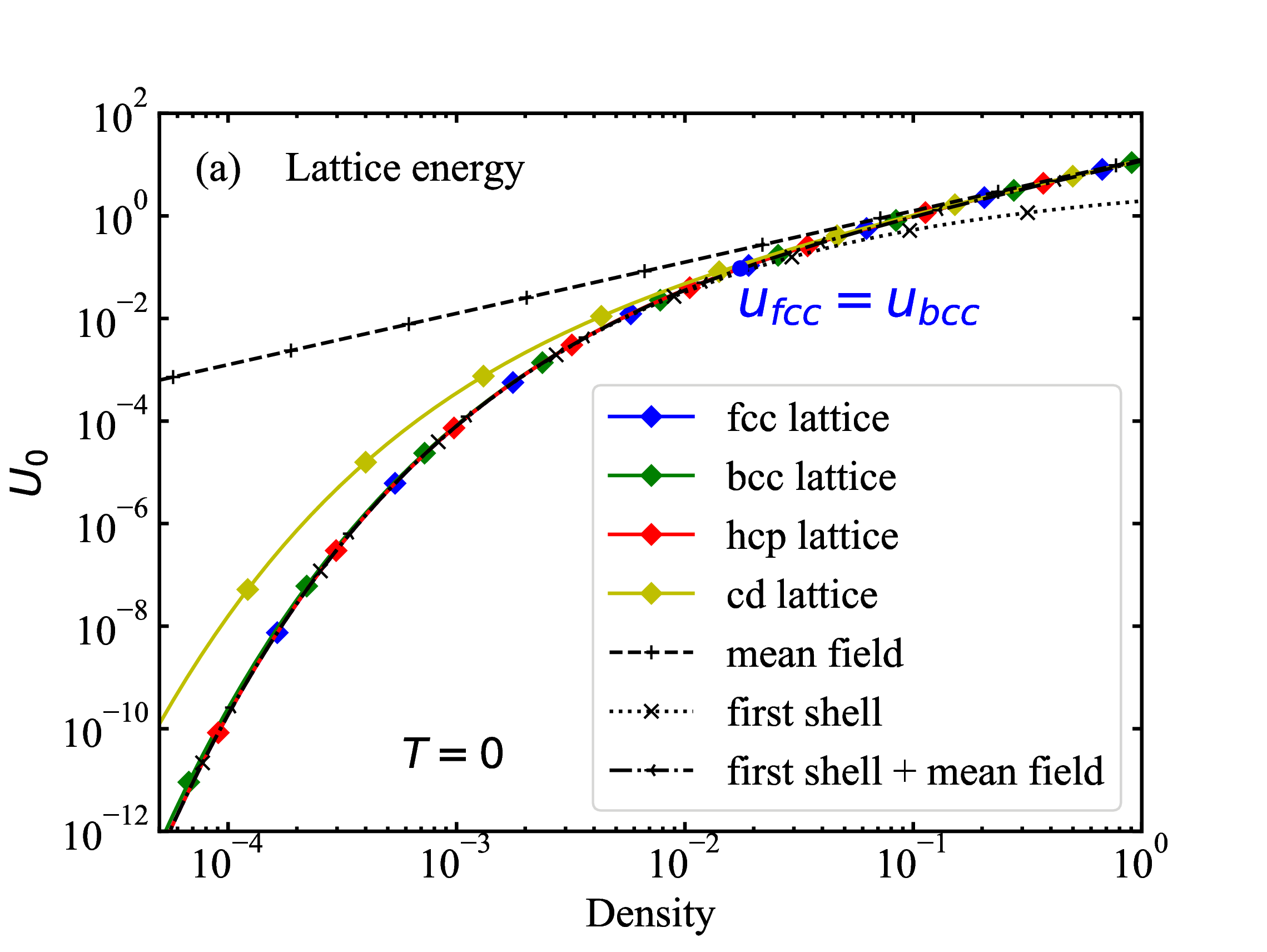 fig/fig1a.pdf
fig/fig1a.pdfRelative difference (fcc and bcc) in lattice energies as function of density at T=0
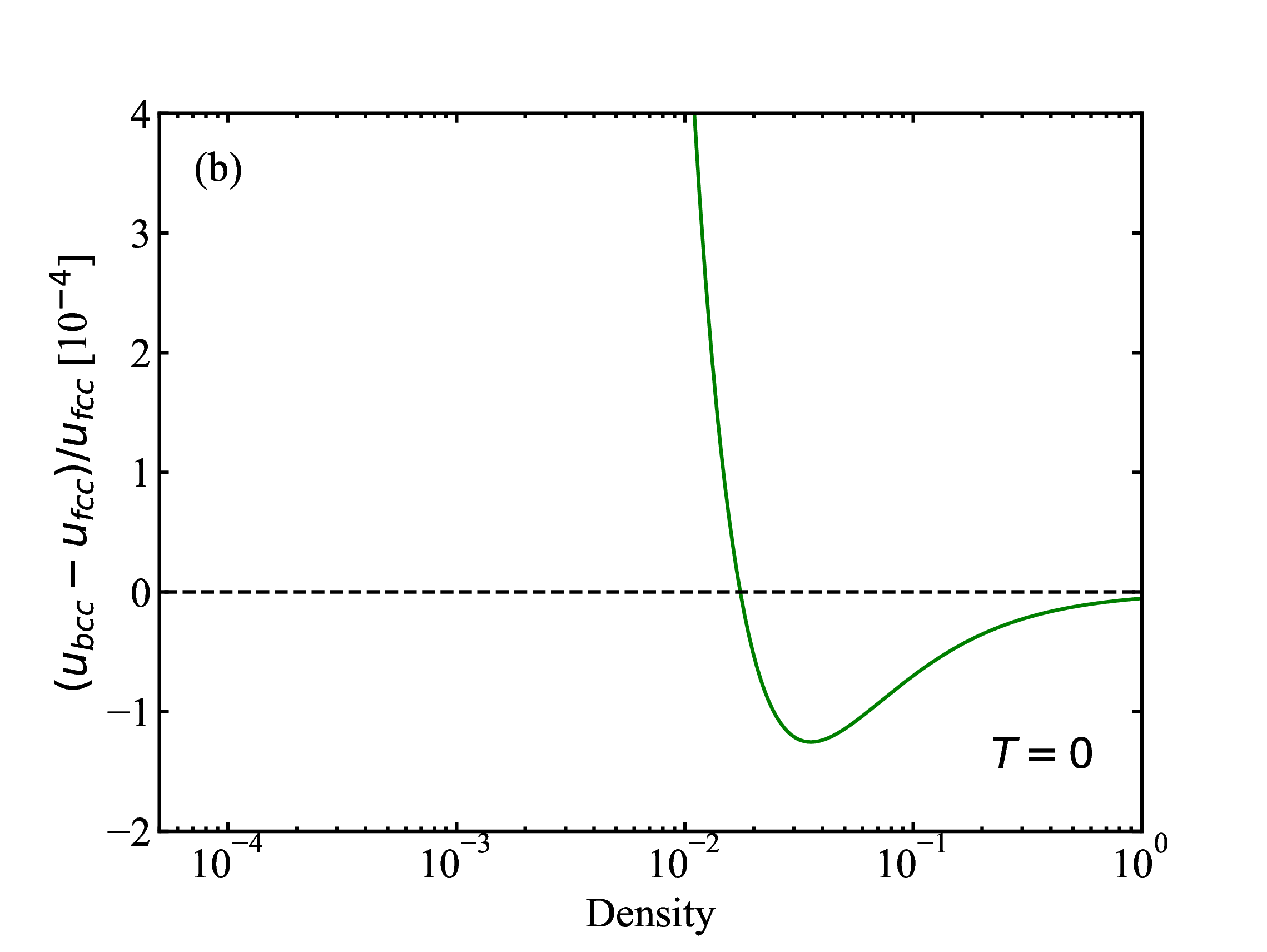 fig/fig1b.pdf
fig/fig1b.pdfRelative difference (fcc and hcp) in lattice energies as function of density at T=0
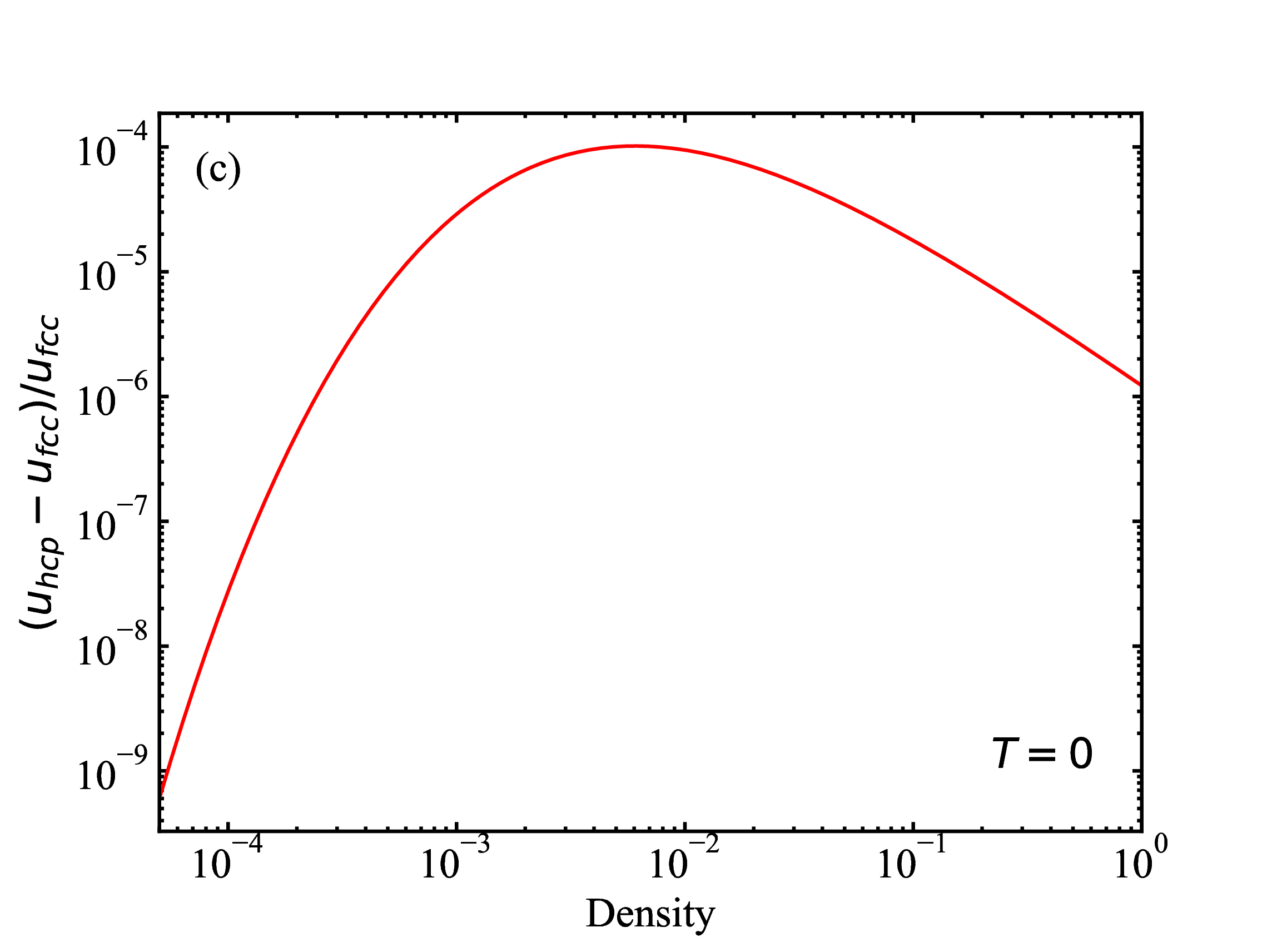 fig/fig1c.pdf
fig/fig1c.pdfRelative difference (fcc and cd) in lattice energies as function of density at T=0
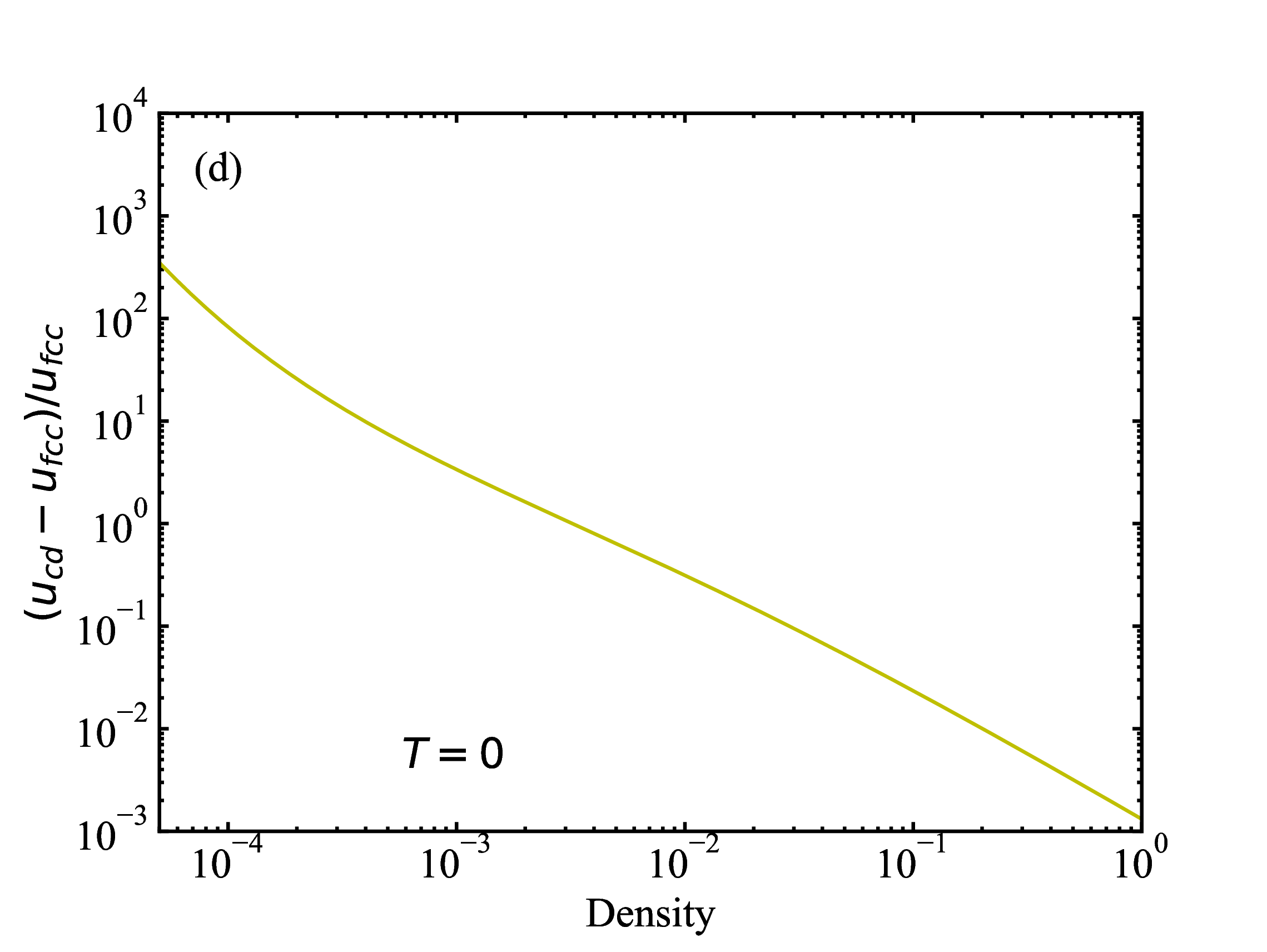 fig/fig1d.pdf
fig/fig1d.pdfRelative difference (fcc and bcc) in chemical potential as function of pressure at T=0
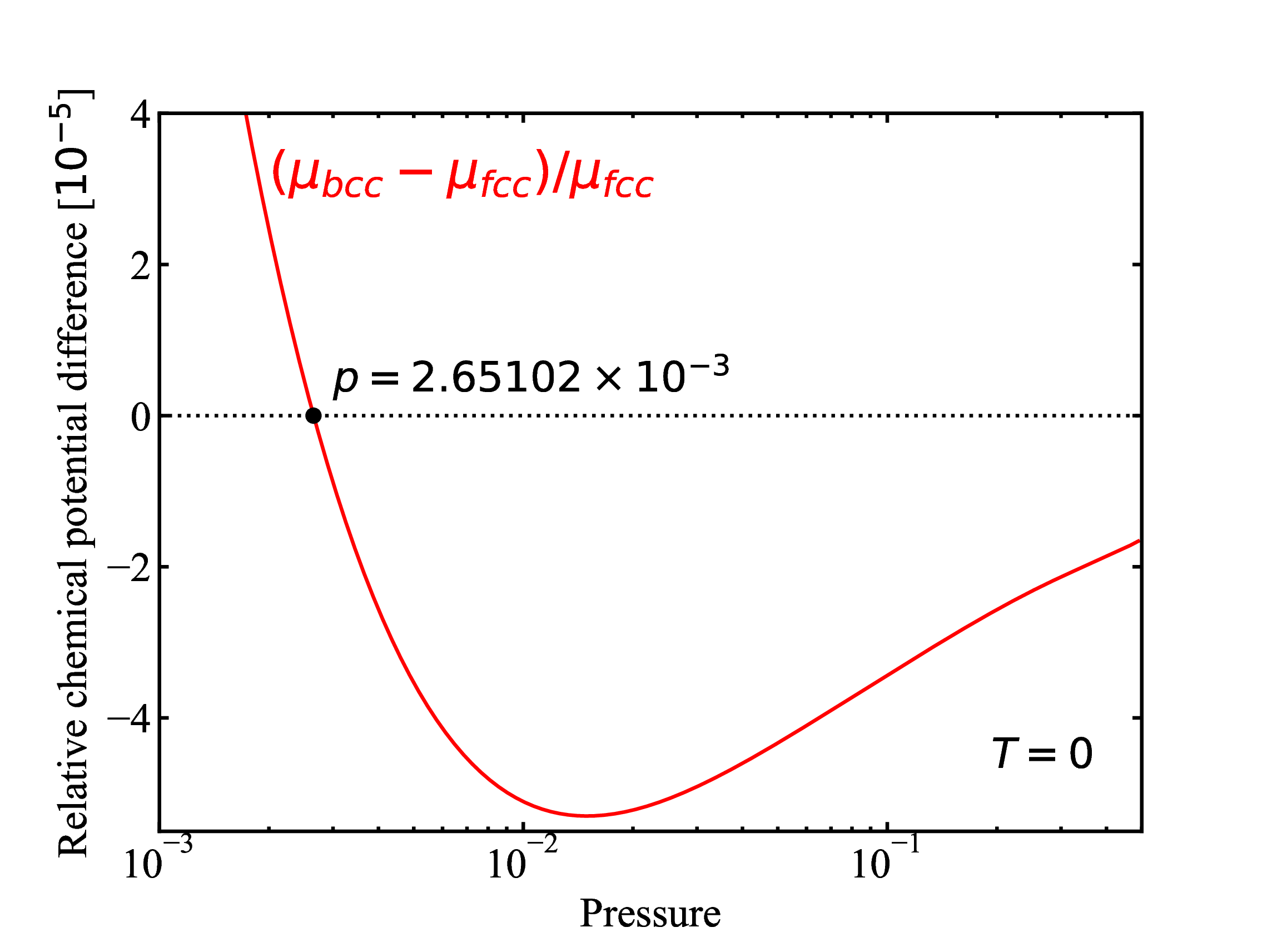 fig/fig2.pdf
fig/fig2.pdfPressure as function of trucation distance
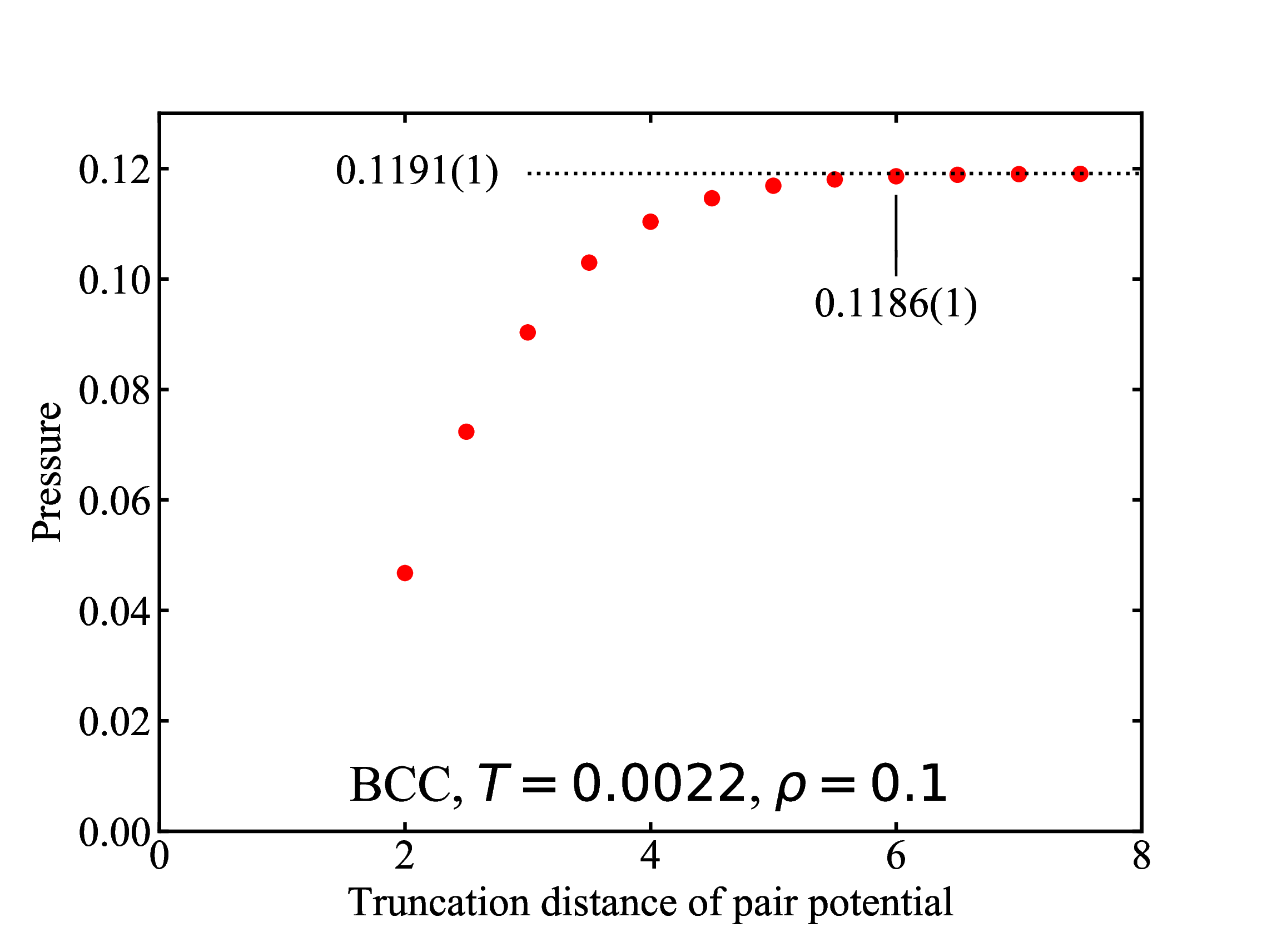 fig/fig3.pdf
fig/fig3.pdfFcc-bcc coexistence from Clausius-Clapeyron integration
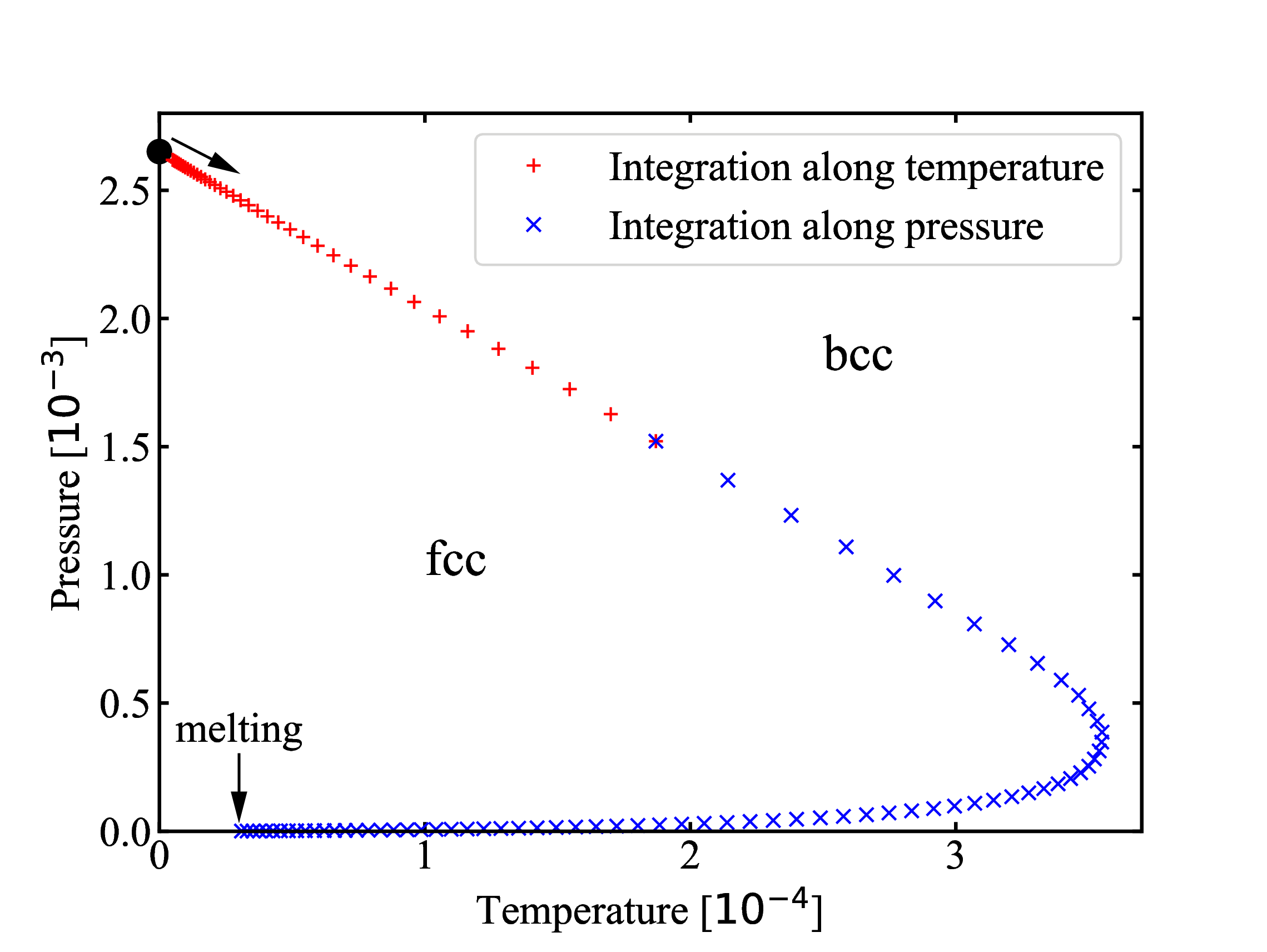 fig/fig4.pdf
fig/fig4.pdfConfiguration from interface-pinning
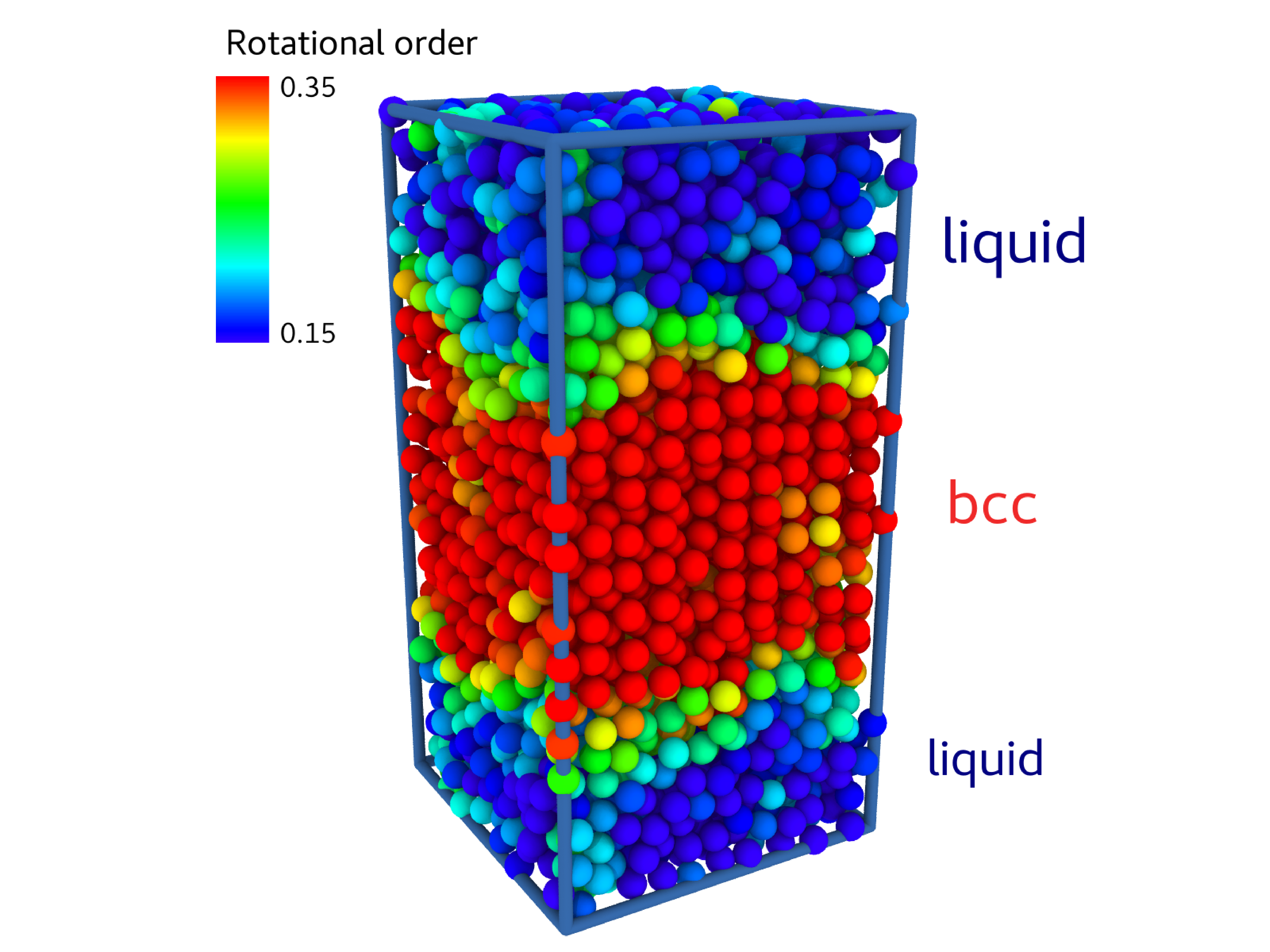 fig/fig5.pdf
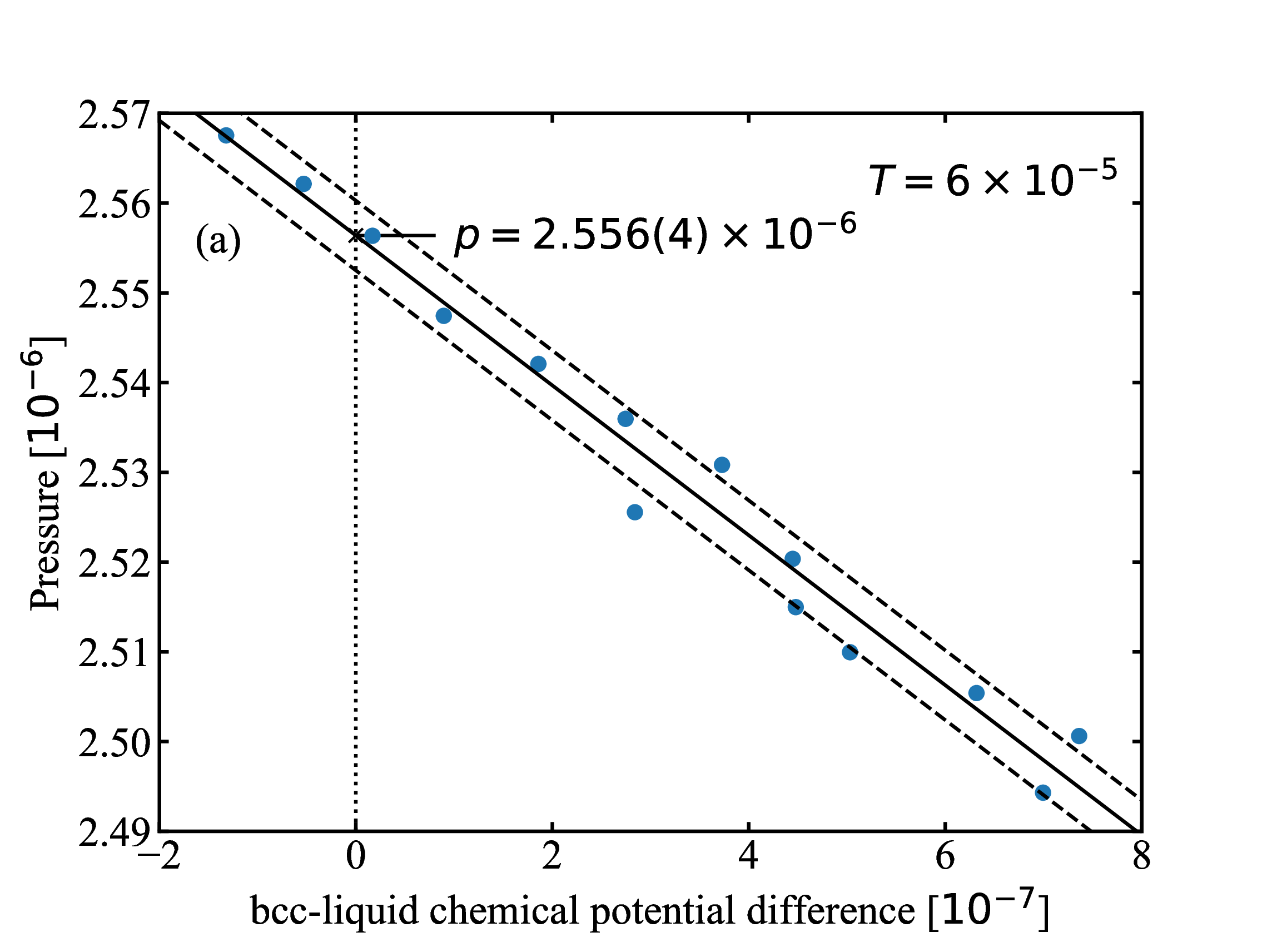
fig/fig5.pdfThe difference in chemical potential (bcc and liquid) from interface-pinning plotted against pressure
 fig/fig6a.pdf
fig/fig6a.pdfRelative difference of the bcc-liquid and fcc-liquid coexistence pressures plotted as a function of temperature
 fig/fig6b.pdf
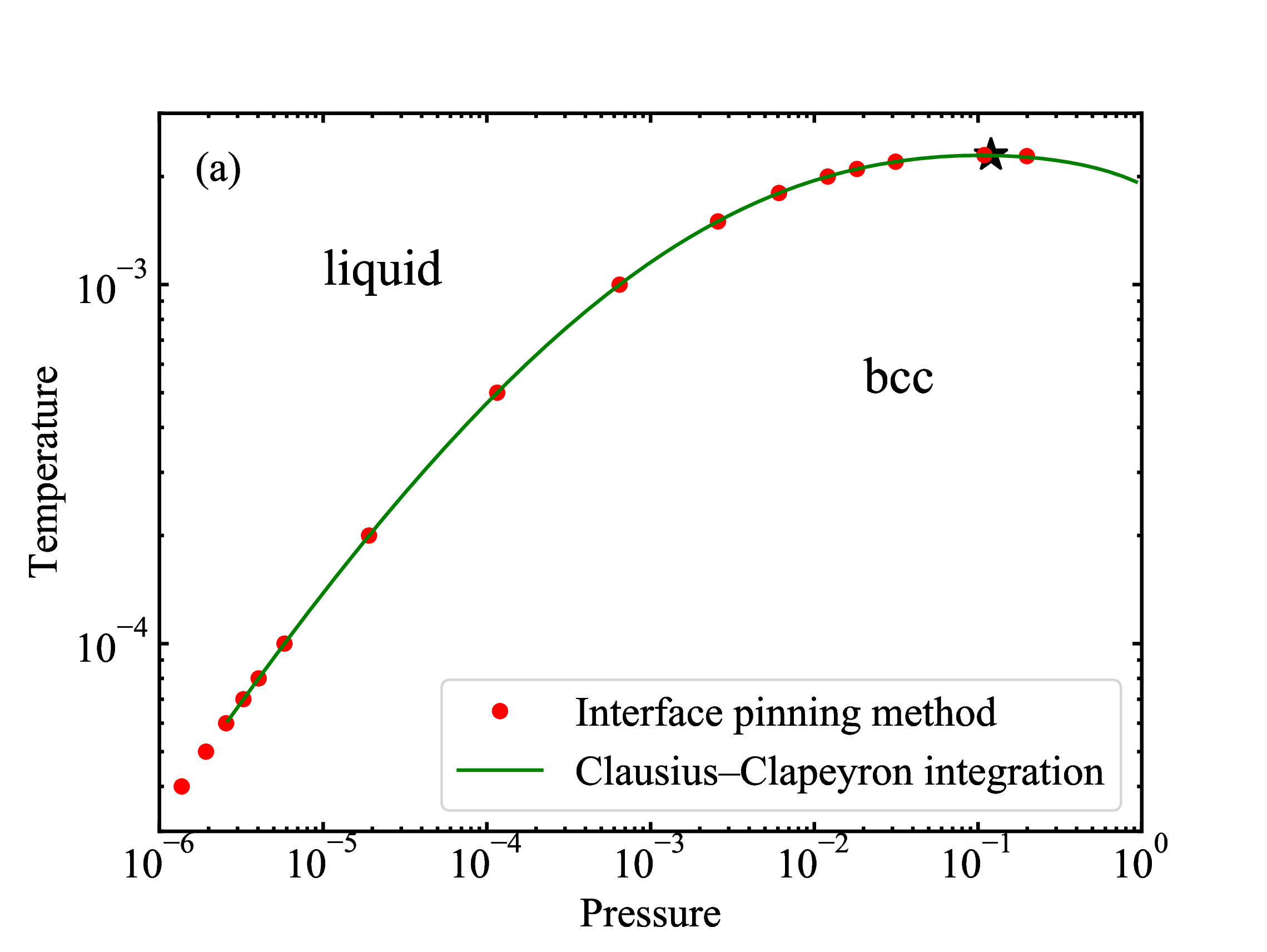
fig/fig6b.pdfbcc-liquid coexistence line
 fig/fig7a.pdf
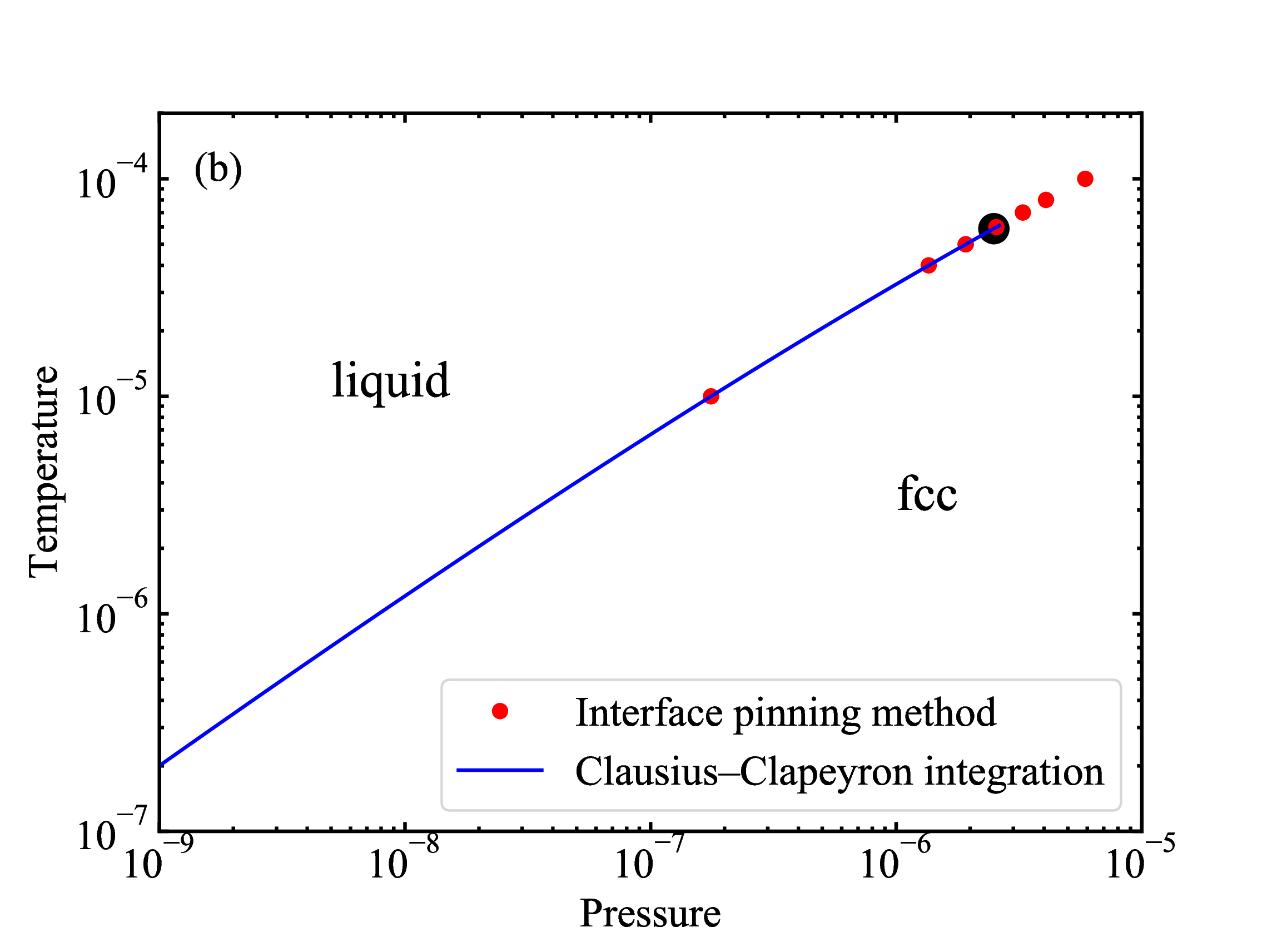
fig/fig7a.pdffcc-liquid coexistence line
 fig/fig7b.pdf
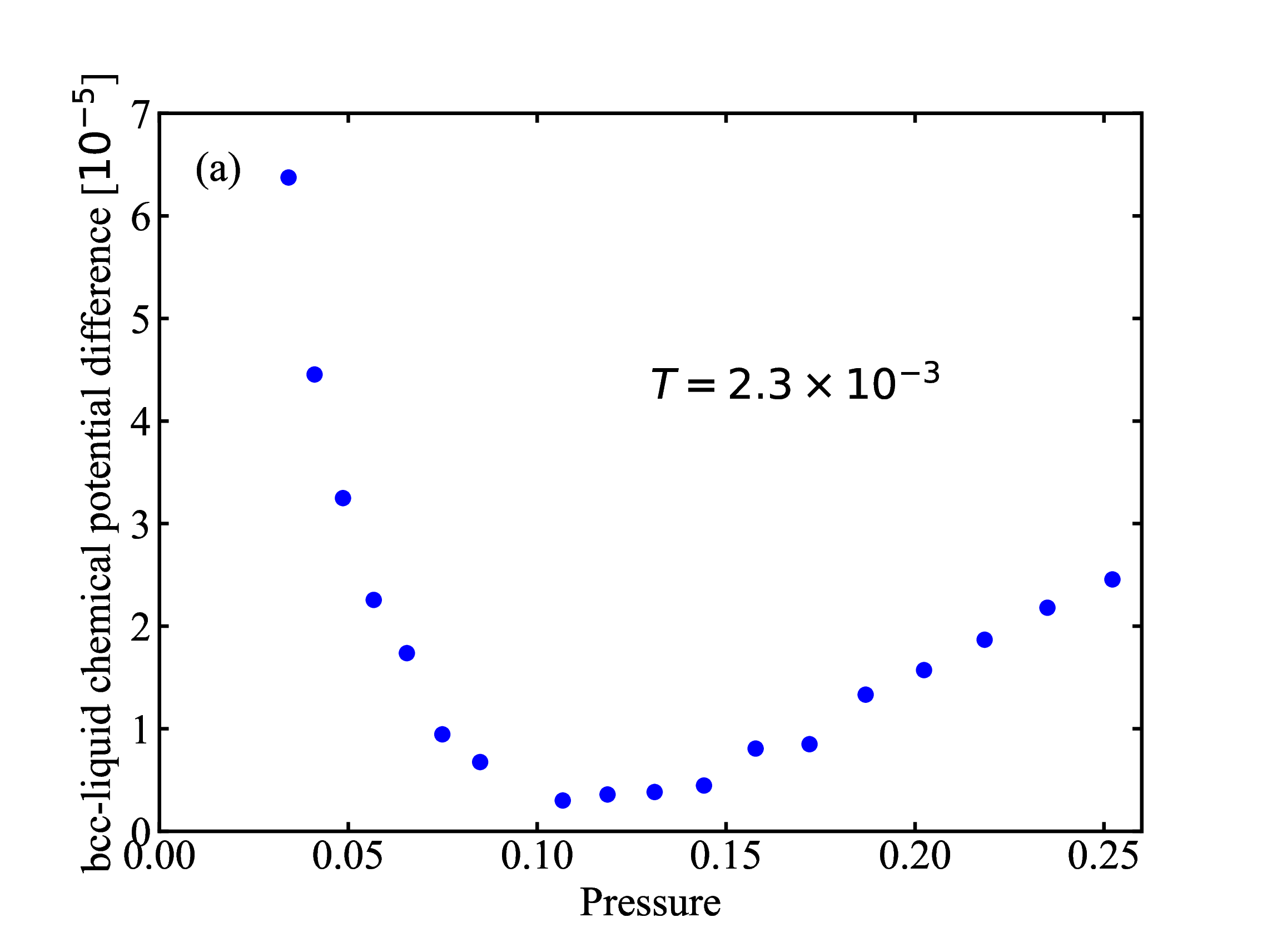
fig/fig7b.pdfDifference in chemical potential (liquid and bcc) plotted as a function of pressure along the T = 2.3 × 10−3 isotherm
 fig/fig8a.pdf
fig/fig8a.pdfEstimated coexistence (melting) temperature plotted as a function of pressure
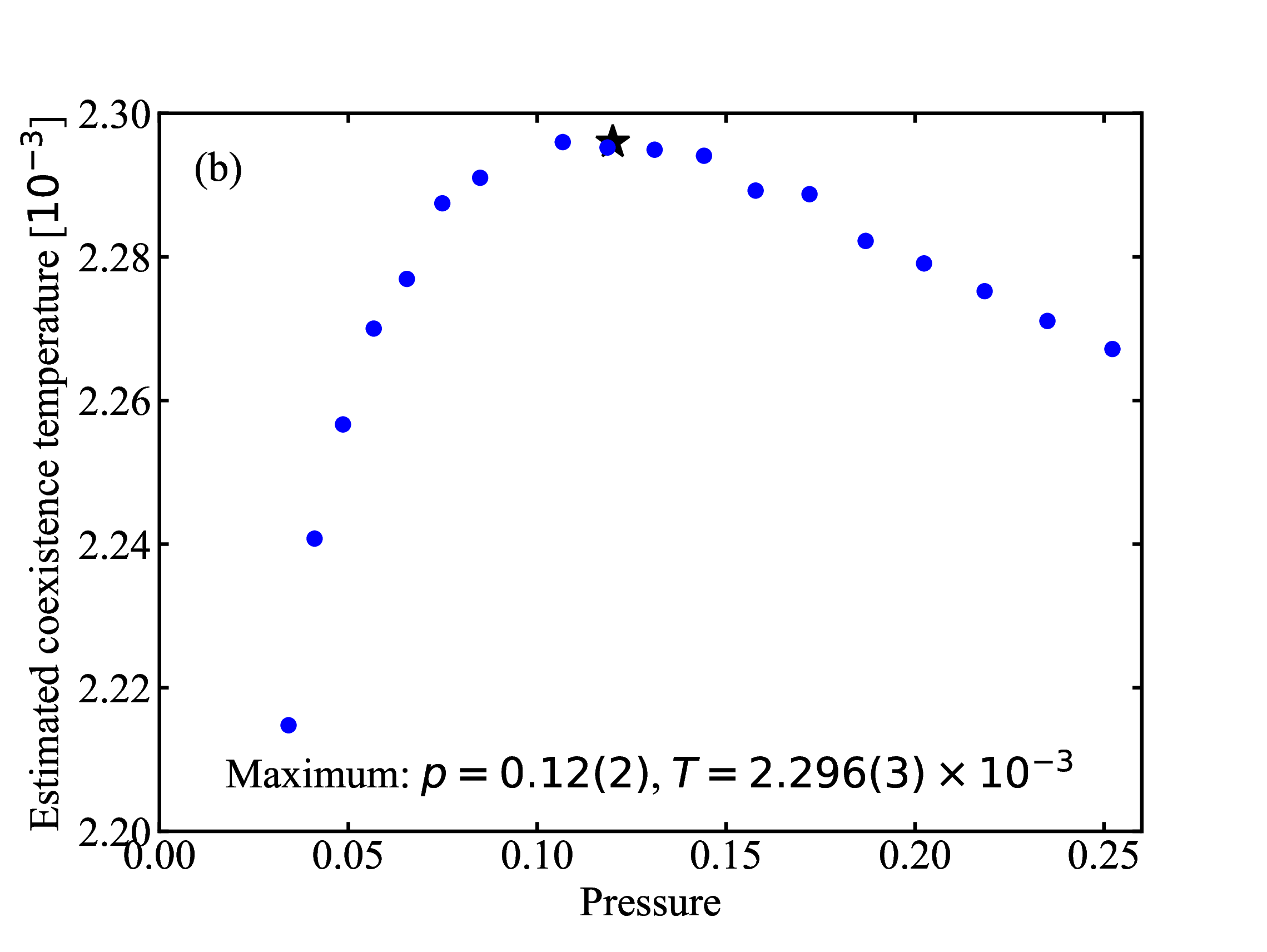 fig/fig8b.pdf
fig/fig8b.pdfPressure-temperature EXP phase diagram
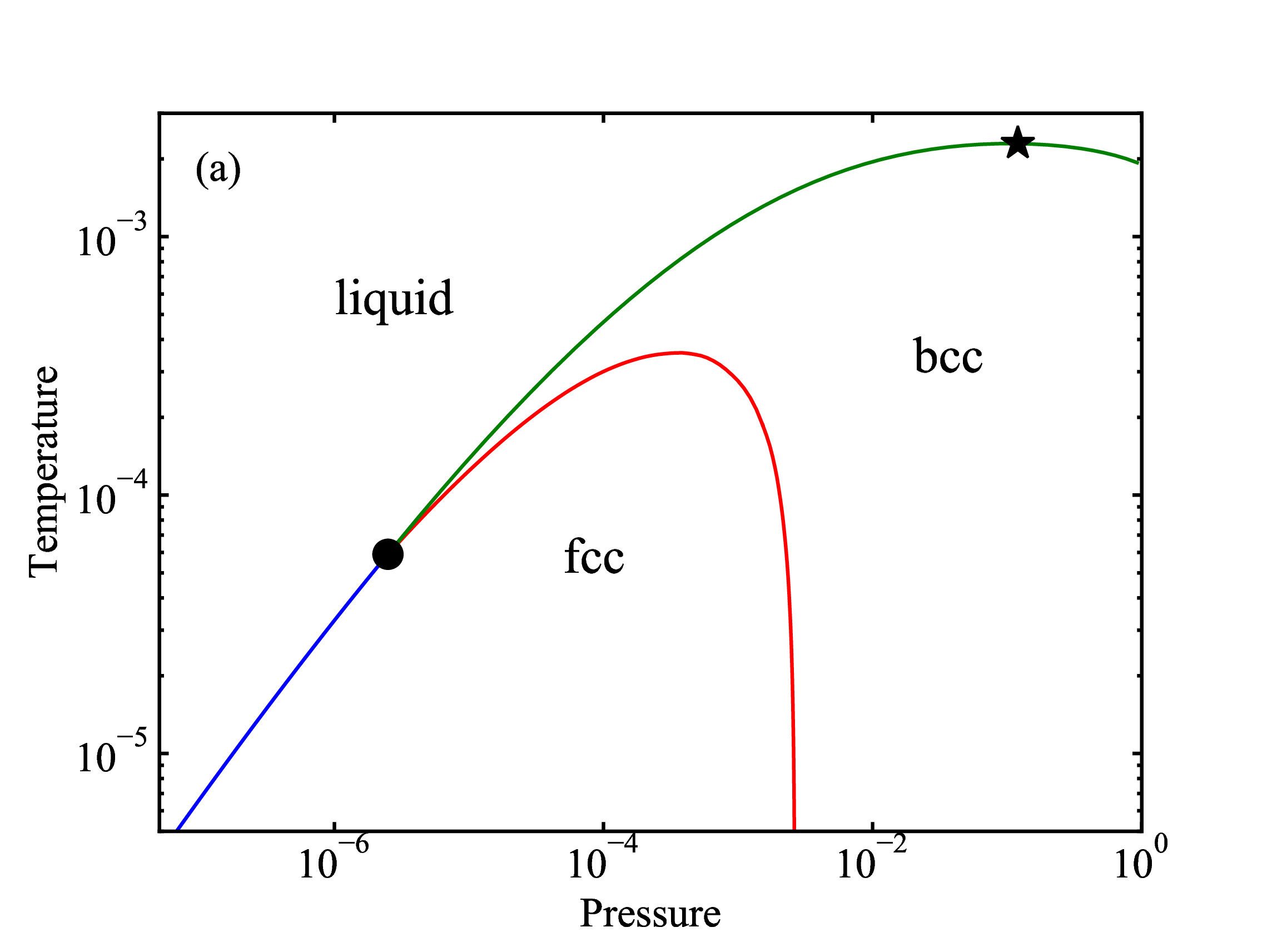 fig/fig9a.pdf
fig/fig9a.pdfDensity-temperature EXP phase diagram
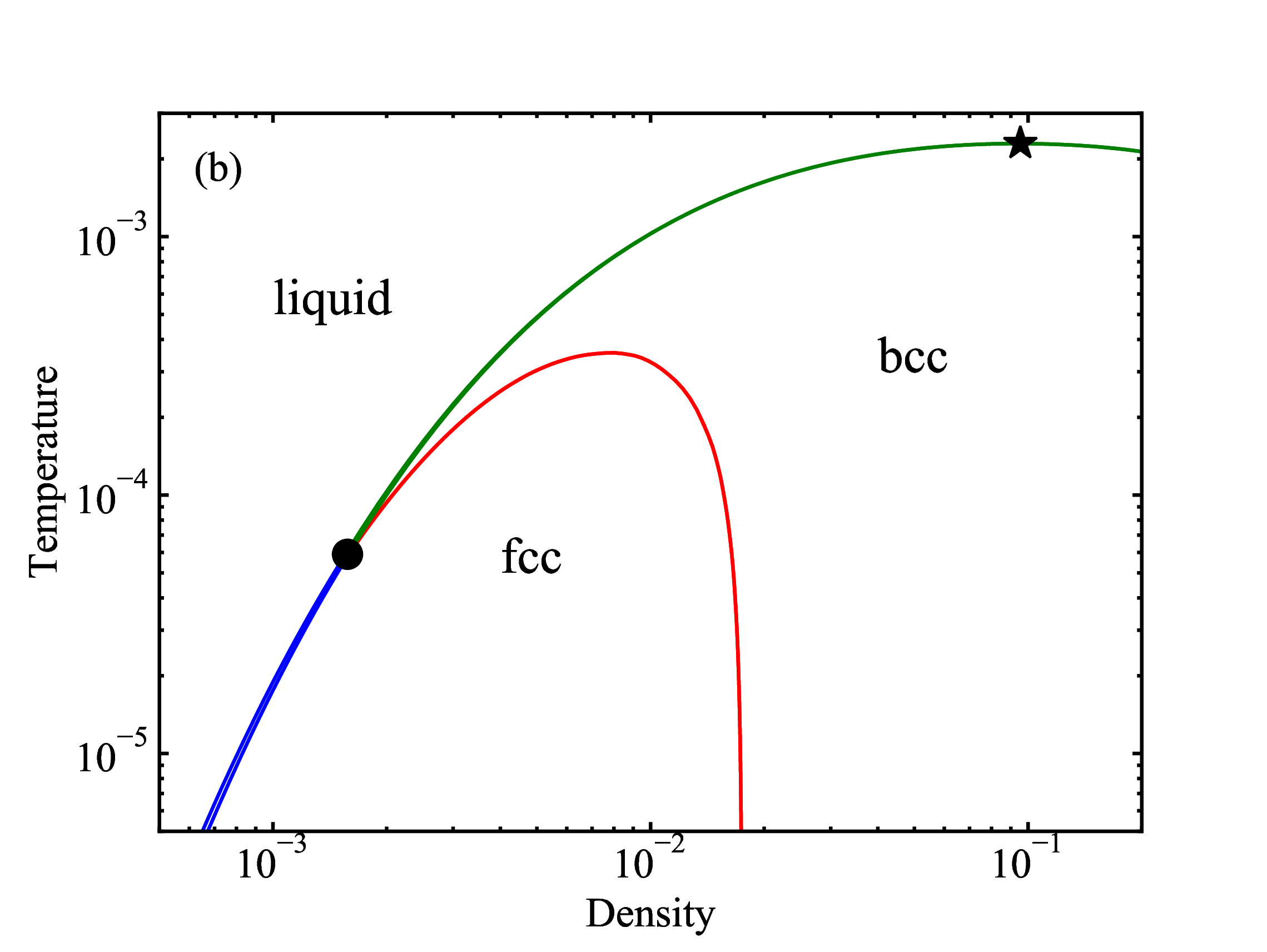 fig/fig9b.pdf
fig/fig9b.pdfPhase-change entropy per particle for the fcc-liquid, bcc-liquid, and fcc-bcc transitions plotted as a function of pressure
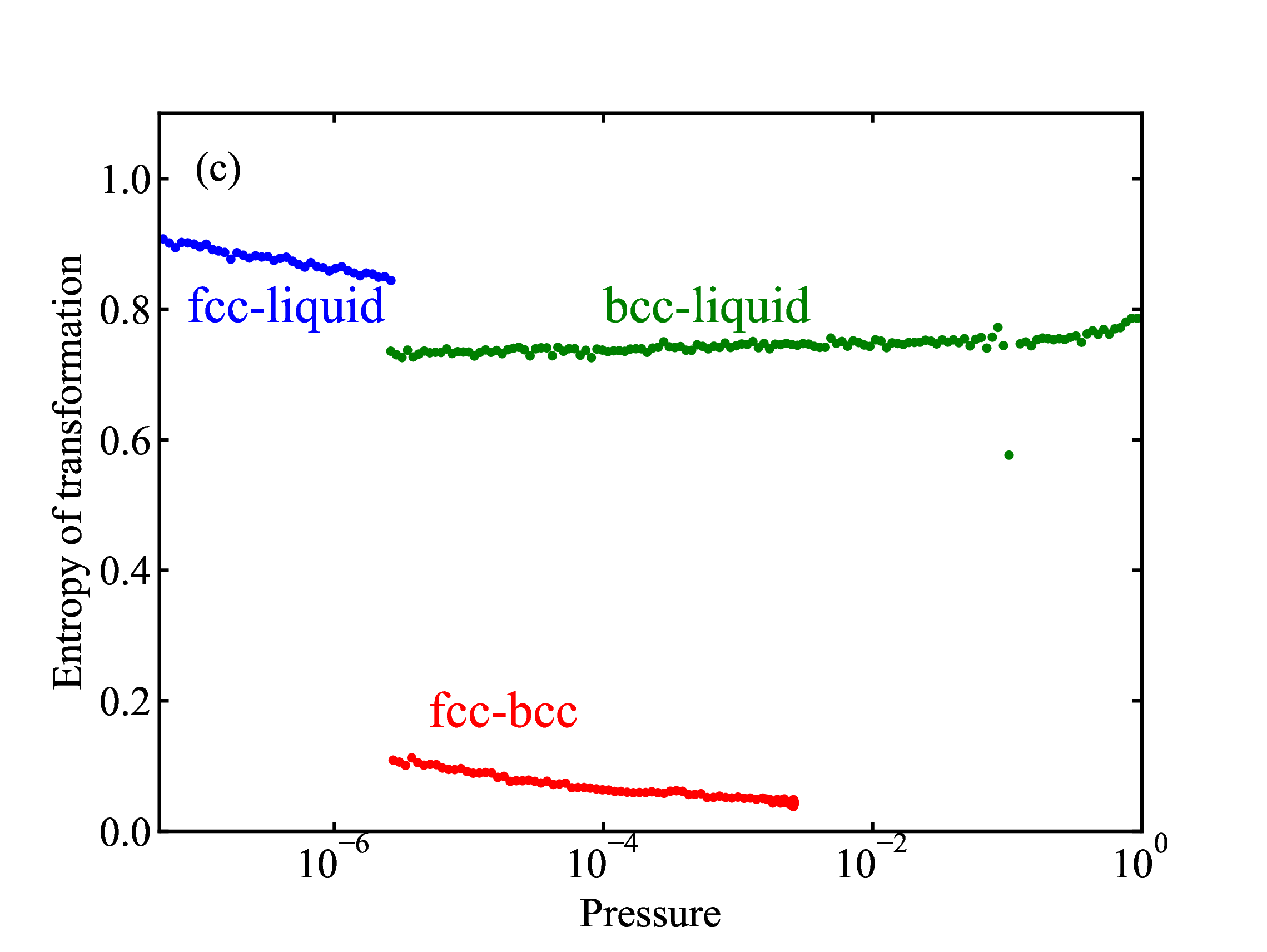 fig/fig9c.pdf
fig/fig9c.pdfRelative density change of the fcc-liquid, bcc-liquid, and fcc-bcc transitions plotted as a function of pressure
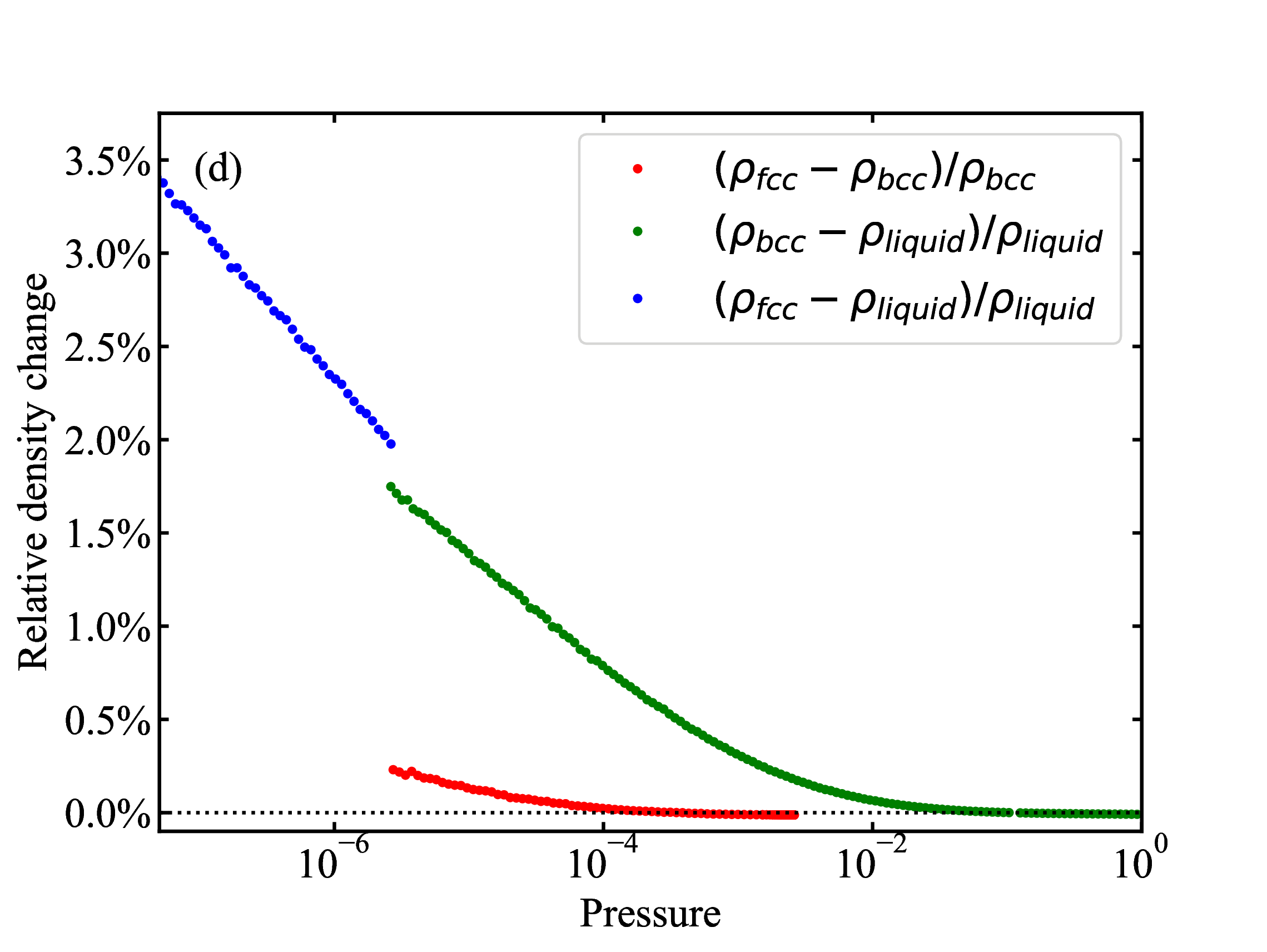 fig/fig9d.pdf
fig/fig9d.pdf
